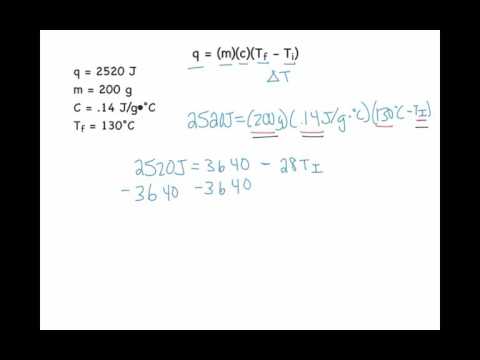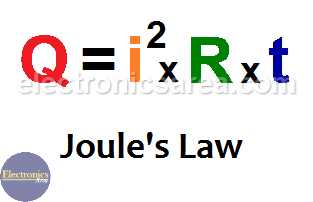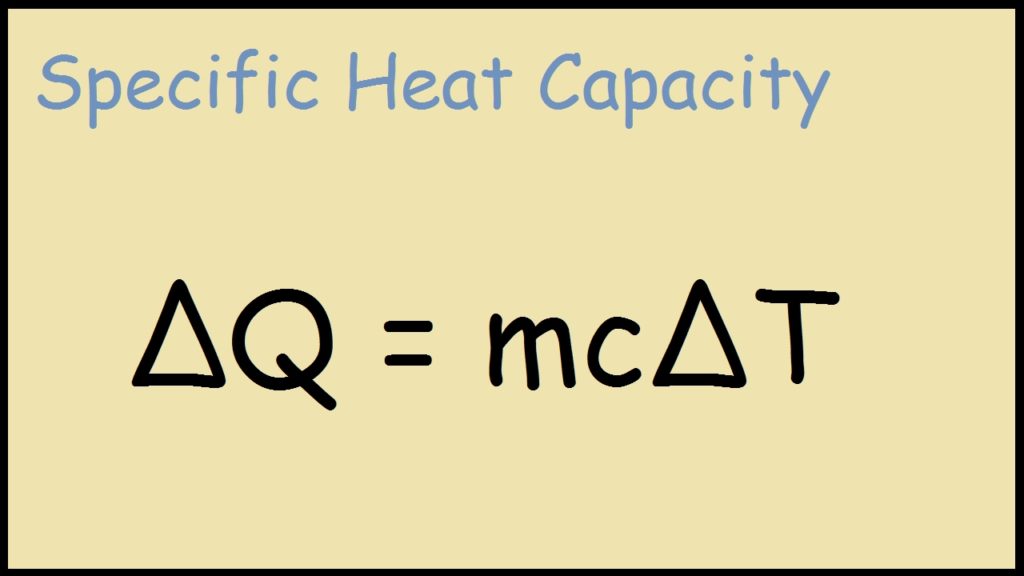SOLVED: 9. A 15.75-g piece of iron absorbs 1086.75 joules of heat energy, and its initial temperature is 25°C. The specific heat capacity of iron is 0.45 J/g°C. What is the final

Question Video: Calculating the Heat Energy Transferred to Water Using Its Specific Heat Capacity | Nagwa

Thermodynamics Ch 10 Energy Sections Thermodynamics The 1st Law of Thermodynamics The Law of Conservation of Energy is also known as The 1st. - ppt download