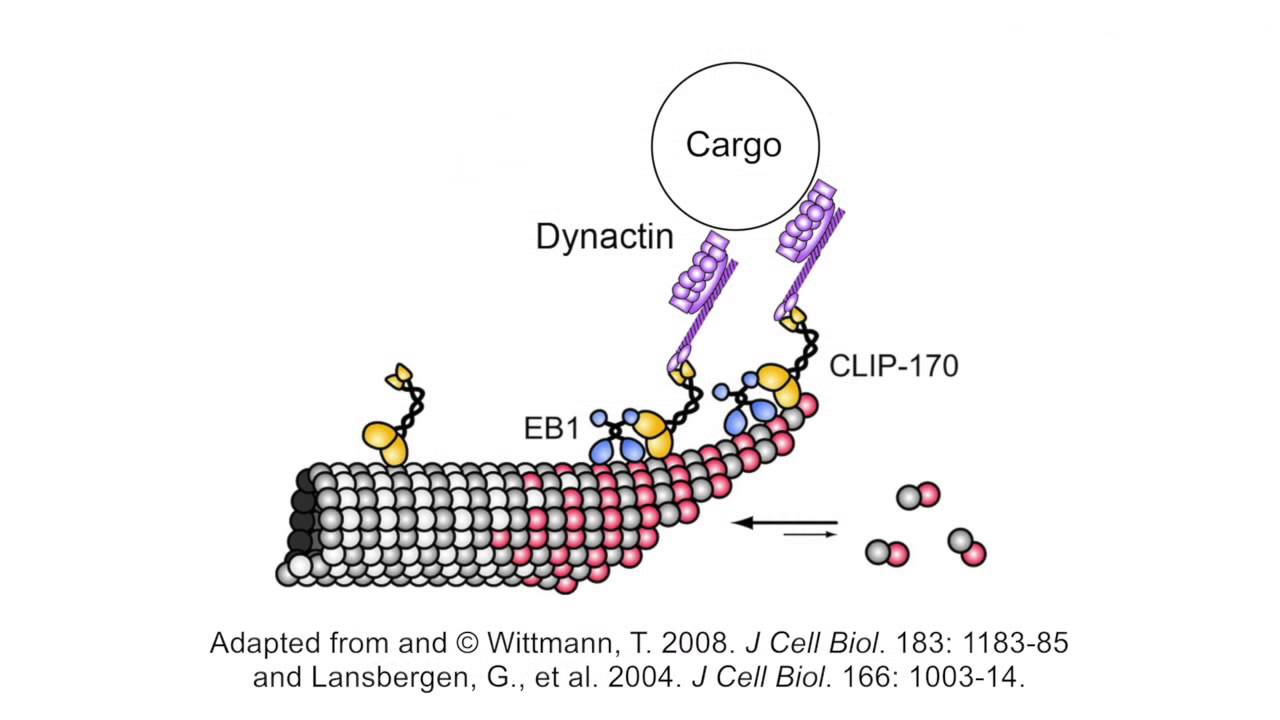
Interactions between CLIP-170, Tubulin, and Microtubules: Implications for the Mechanism of CLIP-170 Plus-End Tracking Behavior | Molecular Biology of the Cell
Selective visualization of growing MT ends with CLIP170. CHO cells were... | Download Scientific Diagram
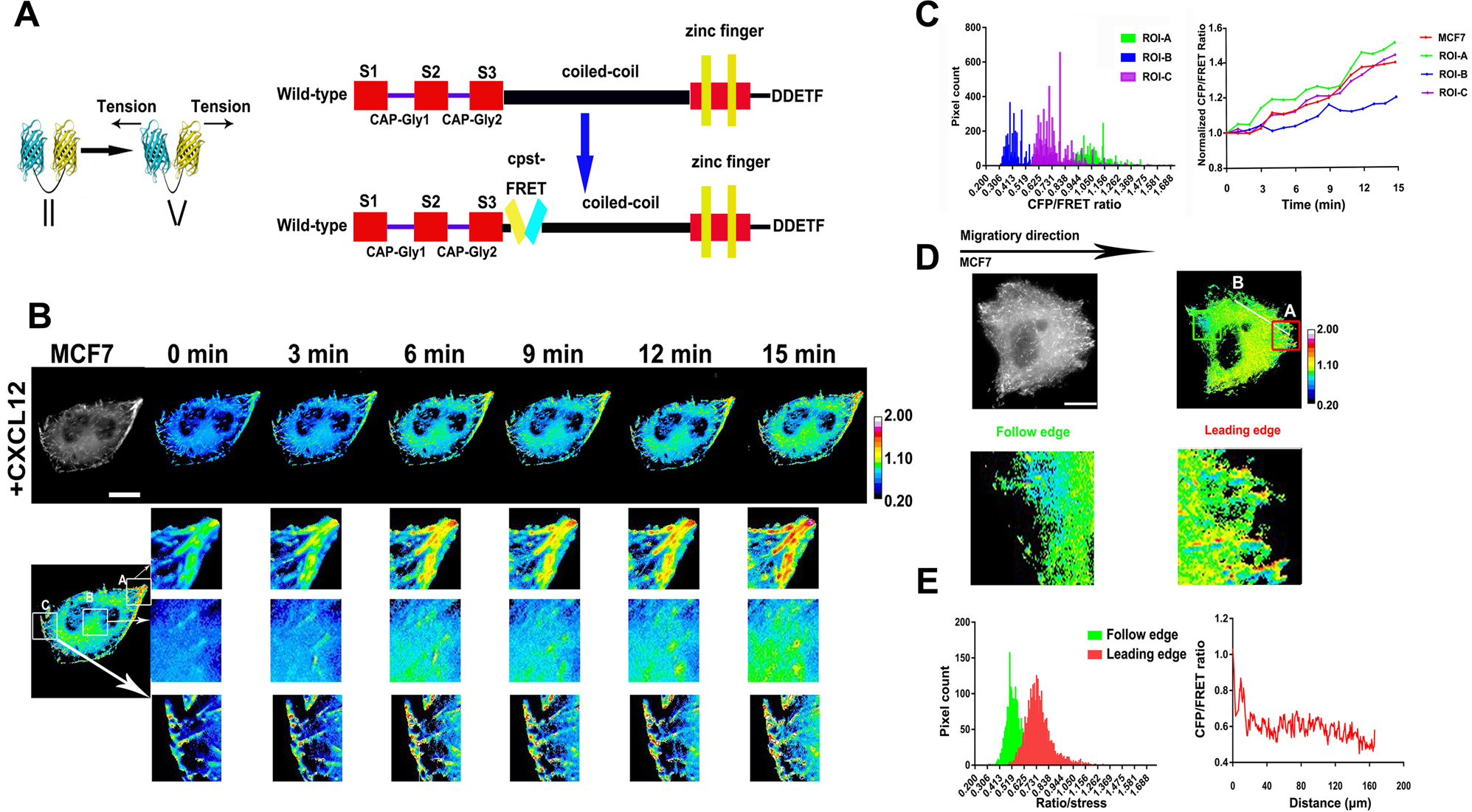
Tension of plus-end tracking protein Clip170 confers directionality and aggressiveness during breast cancer migration | Cell Death & Disease
Subcellular distribution of CLIP-170 throughout the mitotic cycle of... | Download Scientific Diagram

Quelle fonction pour la CLIP-170? : recherche de partenaires et nouveaux outils d'investigation | Semantic Scholar

CLIP‐170 spatially modulates receptor tyrosine kinase recycling to coordinate cell migration - Zaoui - 2019 - Traffic - Wiley Online Library
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular

Conformational changes in CLIP-170 regulate its binding to microtubules and dynactin localization. - Abstract - Europe PMC

Phosphorylation of CLIP‐170 by Plk1 and CK2 promotes timely formation of kinetochore–microtubule attachments | The EMBO Journal

Phosphorylation of CLIP‐170 by Plk1 and CK2 promotes timely formation of kinetochore–microtubule attachments | The EMBO Journal

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology

Ninein is essential for apico-basal microtubule formation and CLIP-170 facilitates its redeployment to non-centrosomal microtubule organizing centres | Open Biology
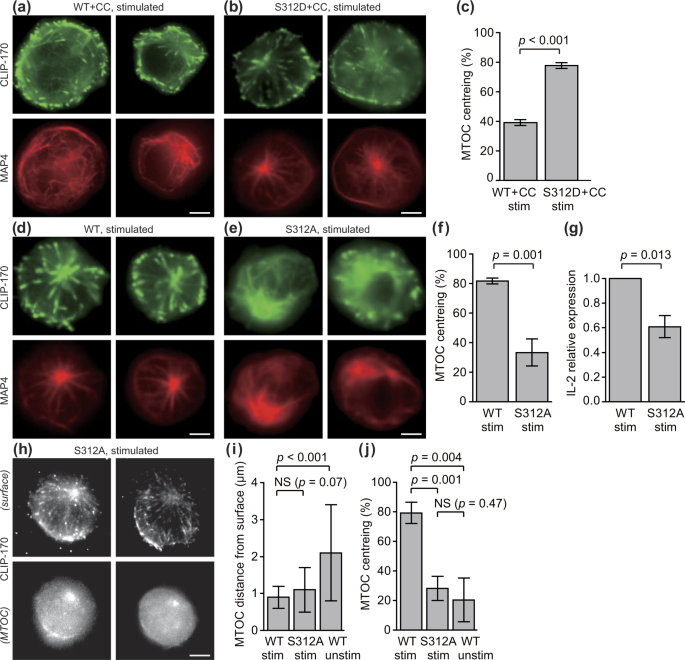
CLIP-170 is essential for MTOC repositioning during T cell activation by regulating dynein localisation on the cell surface | Scientific Reports
Overexpression of the microtubule-binding protein CLIP-170 induces a +TIP network superstructure consistent with a biomolecular condensate | PLOS ONE

α-Tubulin Tyrosination and CLIP-170 Phosphorylation Regulate the Initiation of Dynein-Driven Transport in Neurons - ScienceDirect